热氧化工艺是将硅片暴露于氧化剂和热源的组合环境中,使其形成一层二氧化硅(SiO2)的结果。该二氧化硅层通常是由氢和/或氧元素制成,但也可以使用任何卤素气体。二氧化硅层在环境空气中的晶圆上生长至厚度约为20Å(埃);然而,对于大多数规格来说,热氧化生长二氧化硅会使用热源以催化该反应,并创造出厚度达25,000Å的氧化层。
热氧化晶圆具有诸多应用,但其主要用途是作为电介质材料和MEMS(微机电系统)器件。
在晶圆上进行热氧化有两种主要方式,二者均需要在晶圆表面生长氧气,如图1所示。这与CVD应用不同,CVD应用中的氧化层是沉积在晶圆顶部。
第一种方法是快速热处理(RTP),通常用于薄型干氧氧化,或注入式退火应用。在RTP工艺过程中,晶圆在很短的时间内快速被加热到1000℃以上。曝光几秒后,将晶圆缓慢冷却至室温,以防止开裂。其结果是形成一层薄薄的二氧化硅层,可用于多种应用。
第二种也是更常见的方法是扩散。热氧化扩散发生在约800°和1100°C之间,有两种方法可以生长该薄膜:“湿氧”或“干氧”。
湿氧氧化
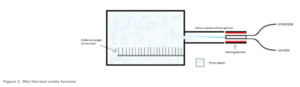
湿氧氧化薄膜一般用于需要较厚的二氧化硅层的应用。为了防止杂质,这些薄膜通常在石英管中生长,同时使用热源和纯蒸汽的组合装置。大多数主要制造商使用安装在石英管中的外部热源,且与高温炉分开,如图2所示。虽然不常见,但有些系统使用内置火焰来代替加热晶圆。在氧化过程中,外部热源的温度升高至800℃以上。这会导致气体自燃,并在没有点火源的情况下产生蓝色火焰。该火焰会产生纯蒸汽,因此被称为湿氧氧化。纯蒸汽通过容纳火焰的管道进入晶圆所在的高温炉。一旦蒸汽进入石英室,它就会膨胀并均匀地分布在整个炉内。
水平放置(如图2所示)或垂直堆叠的晶圆在高温炉中处理数小时,放置时间可根据目标薄膜厚度而改变。二氧化硅的生长不是线性的,因此当晶圆在1,000°C的高温炉中放置处理5个小时后,将形成约10,000Å厚度的氧化层;而如果相同的晶圆在高温炉中放置处理24小时后,将形成约25,000Å厚度的氧化层。第二块晶圆在高温炉中的处理时间长了近五倍,但氧化层厚度只有前一块晶圆的两倍左右。这是因为随着氧化层的生长,氧穿透器件层并与硅衬底相互作用以生成SiO2变得更加困难。
干氧氧化
干氧氧化产生的二氧化硅层比湿氧氧化要薄得多,而且整个过程耗时更长。由于这些限制,该工艺生长的二氧化硅层厚度不超过1,000Å。
干氧氧化生长过程与湿氧氧化生长过程非常相似。唯一的区别是,该工艺使用分子氧而不是纯蒸汽来创建器件层。这可以制造出高均匀度、高介电强度和高密度的二氧化硅晶圆。
厚热氧化物
厚氧化物是指厚度大于2μm(20000Å)的二氧化硅层。
当需要更厚的氧化层时,通常采用湿氧氧化法进行生长。这种技术的生长速度比干氧氧化法快得多,并且可以形成更厚的二氧化硅薄膜。
厚氧化物应用:
湿氧氧化物通常被用作支撑衬底和承载晶圆之间的厚膜绝缘体上硅晶圆的掩膜层。它是用于磨合工具和硅片管脚的化学机械平坦化(CMP)的耗材。
颗粒敏感型氧化物
颗粒敏感型氧化物存在于基板上生长热氧化物的过程中,同时在生长过程中添加最少量的颗粒。为保护晶圆不受颗粒的影响,所有的工艺流程都在超净间中进行。初制晶圆也必须具有较低的颗粒数,并采用原厂包装,以尽量减少颗粒污染。
为了生长这种涂层,晶圆在石英炉内经历了湿氧或干氧热氧化工艺处理。至关重要的是要使用干净的石英炉以保护晶圆免受周围环境中的颗粒影响。

硅谷地区4小时送达

美国境内1天内送达

全球范围3天送达